IDSA, ASTMH release leishmaniasis guidelines following increase in US cases
- Todd MacLaughlan

- Nov 23, 2016
- 4 min read
An increase in leishmaniasis cases among travelers, immigrants and soldiers in the United States prompted the Infectious Diseases Society of America and the American Society of Tropical Medicine and Hygiene to develop new evidence-based recommendations.
“Leishmaniasis is an increasingly common infection in ecotourists traveling to Central and South America. Travelers visiting the jungle in the Amazon basin have a high likelihood of being exposed,” Naomi E. Aronson, MD, of the Uniformed Services University of the Health Sciences in Bethesda, Maryland, said in a press release. “The cutaneous and mucosal forms of the infection cause serious scarring and visceral leishmaniasis can be deadly, so timely diagnosis and treatment managed by an infectious disease physician is vital.”

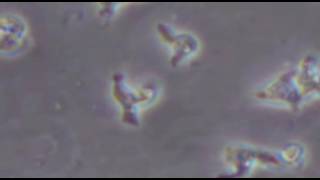
Leishmaniasis is a parasitic disease that is transmitted through the bite of infected sand flies. There are more than 20 Leishmania species that are pathogenic for humans, according to Aronson and colleagues. The parasites are prevalent in more than 90 countries, including Central and South America, Asia, Africa, the Middle East and southern Europe. In the United States, several autochthonous cases have been reported in Texas and Oklahoma; however, almost all leishmaniasis cases in North America occur among immigrants, international travelers and military personnel returning from endemic regions.
The most common clinical syndromes of Leishmania are cutaneous leishmaniasis (CL), mucosal leishmaniasis (ML) and visceral leishmaniasis (VL). The CDC estimates that 700,000 to 1.2 million cases of CL are reported each year. Skin lesions caused by CL are often painless and may heal on their own. In some cases, Leishmania can spread from the skin to the nasooropharyngeal mucosa and cause ML. The disease may damage nasal and pharyngeal structures and could lead to death due to complicating aspiration pneumonia. VL is caused by systemic infection and may be fatal if left untreated. According to the CDC, 200,000 to 400,000 new cases of VL occur each year.
Diagnosing leishmaniasis can be challenging because the clinical manifestations of disease vary and may appear more than a month after exposure. Furthermore, inconsistent study data, the incidence of recurrent infections and limited treatment options make leishmaniasis difficult to treat. The only FDA–approved therapies are IV liposomal amphotericin B (L-AmB) for VL, heat treatment with the ThermoMed device (Thermosurgery Technologies Inc.) for CL, and Impavido (miltefosine; Knight Therapeutics Inc.) for specific cases of CL, ML and VL. Although these therapies are more than 80% effective, they don’t always eradicate the parasite and additional treatment may be necessary, according to the press release. There is no definitive method to parasitologic cure for leishmaniasis; therefore, the IDSA and ASTMH recommend that patients with prior infection avoid donating blood. In addition, a patient’s history of leishmaniasis should be considered before organ and tissue donations.
Since there are no vaccines or preventive medications for leishmaniasis, travelers visiting endemic areas are urged to wear protective clothing, apply insect repellents containing DEET and use insecticide-impregnated bed nets. If a patient is suspected of having CL, health care providers can use PCR testing or culture to identify the type of Leishmania and determine how to treat the infection. If VL is suspected, physicians can use a rapid diagnostic blood test, rK39 (Kalazar Detect, InBios), and confirm the results with PCR or culture.
According to the guidelines, published in Clinical Infectious Diseases, systemic therapy is recommended for patients with complex CL lesions who are at risk for ML, as well as those with ML and abnormalities compatible with VL. The agent, dose and duration of therapy should be individualized based on the parasite species and host factors. There is no ideal therapy for CL. ML, however, is traditionally treated with a pentavalent antimonial (SbV) compound (20 mg SbV/kg daily IV or intramuscular for 28 to 30 days) or amphotericin B deoxycholate (0.5-1.0 mg/kg IV daily or every other day for a cumulative total of 20-45 mg/kg). More recently, the armamentarium for ML has expanded to include L-AmB (cumulative dose of 20-60 mg/kg) and oral miltefosine (approximately 2.5 mg/kg for 28 days). Patients with VL are recommended to receive 3 mg/kg of L-AmB intravenously on days 1 to 5, 14 and 21; however, certain patients can receive 2.5 mg/kg of oral miltefosine for 28 days. Leishmaniasis treatments vary among immunocompromised patients, such as those with HIV, lymphatic or hematological malignancies, as well as other special populations, including pregnant or lactating women, young children and older adults.
“In general, clinically manifest cases of VL and ML should be treated even in these special populations of persons because the benefits of treatment typically outweigh the risks,” Aronson and colleagues wrote. “Decisions regarding whether and how to treat cases of CL in persons with special characteristics or comorbid conditions should take into account the potential risks and benefits of various approaches, such as initially observing without antileishmanial therapy, deferring treatment, and using local therapy as the sole approach or as a temporizing measure, if otherwise appropriate and feasible.” – by Stephanie Viguers
Disclosure: Aronson received payments from Up to Date. Please see the full study for a list of all other authors’ relevant financial disclosures.


























Hôm qua mình đang tìm thông tin về mấy trò chơi mới, tình cờ thấy có nhắc tới tg88x.com trong một bài viết. Mình vào xem thử cho biết, thấy giao diện khá đơn giản nhưng dễ nhìn, không bị rối mắt. Tốc độ tải trang cũng nhanh, điều này làm mình hài lòng. So với những trang khác mà mình hay vào, mình thấy tg88x.com có phần dễ sử dụng hơn, dễ dàng tìm kiếm thông tin cần thiết. Mình có bookmark lại để sau này quay lại xem thêm, vì thấy có vẻ có nhiều nội dung thú vị. Thực sự là một trải nghiệm khá ổn!
MM88 là nhà cái trực tuyến uy tín hàng đầu Việt Nam, được mm888 media giới thiệu là nền tảng cá cược minh bạch, công bằng và chuyên nghiệp. Giao diện hiện đại, thao tác mượt mà, bảo mật cao giúp người chơi an tâm tuyệt đối.
The learning curve in Poor Bunny is perfect—easy to pick up, but it takes a lot of practice to get really good.
Exploring the Rise of Live Dealer Games: A Trend for 2025
The online gambling industry is in the midst of a profound transformation, with live dealer games emerging as one of the most exciting and game-changing trends set to define 2025 and beyond. Blending cutting-edge technology with the authenticity of real-world casino interaction, live dealer games bridge the gap between virtual convenience and human connection. Players can now enjoy real-time gameplay streamed from professional studios, complete with live chat features that foster community and engagement. To experience this new era firsthand, platforms like https://casinosanalyzer.com/online-casinos/megamedusa.com serve as valuable resources. CasinosAnalyzer is a community of professional gamblers offering transparent reviews and insights on platforms such as MegaMedusa Casino — assessing their live dealer…
Experience passion and romance with our Call Girls in Delhi. Our professional Delhi Call Girls Service is designed for men who seek luxury and intimacy. Book discreetly with our Delhi Call Girl Service and indulge in the most exciting Delhi Call Girls Service.